We’re taking a look at the four major entry routes through which a chemical can enter the body. In our last blog, we unpacked the ways a person can inhale chemicals, and the respiratory protection your workplace must have in place. Today, we review how toxins and hazardous chemical substances are absorbed through the skin.
Did you know? Chemicals that pass through the skin are nearly always in liquid form
Solid chemicals and gases or vapours do not generally pass through the skin unless they are first dissolved in moisture on the skin's surface.
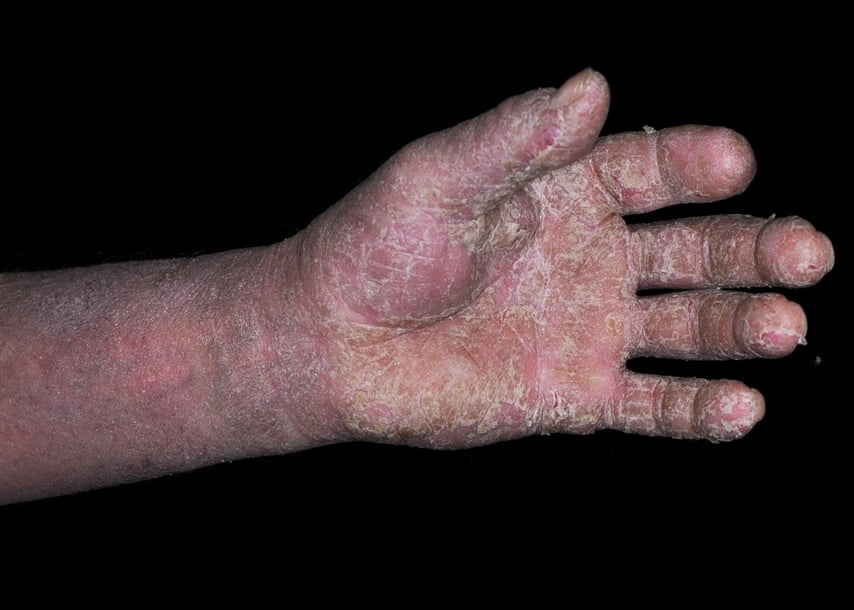
Occupational skin diseases (OSDs) are the second most common type of occupational disease and can occur in several different forms including:
- irritant contact dermatitis
- allergic contact dermatitis
- skin cancers
- skin infections
- skin injuries, and
- other miscellaneous skin diseases.
Contact dermatitis is one of the most common types of occupational illness, with estimated annual costs exceeding $1 billion.
9 industries most at risk of potentially harmful exposures of the skin
- Food service
- Cosmetology
- Health care
- Agriculture
- Cleaning
- Painting
- Mechanics
- Printing/lithography
- Construction
|
A quick biology lesson The skin consists essentially of two layers: a thin, outer layer called the epidermis and a much thicker layer underneath, called the dermis. The epidermis consists of several layers of flat, rather tightly-packed cells, which form a barrier against infections, water, and some chemicals. This barrier is the external part of the epidermis. It is called the keratin layer, and is largely responsible for resisting water entry into the body. It can also resist weak acids but is much less effective against organic and some inorganic chemicals. The keratin layer contains fat and fat-like substances that readily absorb chemicals that are solvents for fat, oil, and grease. |
Organic and alkaline chemicals can soften the keratin cells in the skin and pass through this layer to the dermis, where they are able to enter the bloodstream. Areas of the body such as the forearms (which may be particularly hairy), are most easily penetrated by chemicals as they enter through the small duct containing the hair shaft. Chemicals can also enter through:
- cuts
- punctures, or
- scrapes of the skin
since these are breaks in the protective layer.
Contact with some chemicals, such as detergents or organic solvents, can cause skin dryness and cracking. There can also be:
- hives
- ulcerations, or
- skin flaking.
All these conditions weaken the protective layer of the skin and may allow chemicals to enter the body.
Chemicals can vary enormously in the degree to which they penetrate the skin:
- Some solvents may soften the keratin layer but are not believed to penetrate much further unless there is prolonged skin contact.
- Other chemicals can readily pass through the epidermis and subsequently enter the bloodstream.
- Some chemicals are so corrosive they burn holes in the skin, allowing entry for infection or other chemicals.
In some instances, chemicals may enter the body by accidental injection through the skin. This situation may occur in hospital settings or in industrial hole-punching or injection processes. Once in the bloodstream, these chemicals can be transported to any site or organ of the body where they may exert their effects.
Skin protection at work
The most common protection for the skin is PPE, and specifically gloves (as the most used body part during the course of performing our work, our hands are most likely to be exposed to toxins). Just as no one type of respirator can protect from all air contaminants, no glove can protect from all substances and tasks that could injure the hand.
Like most PPE, gloves need to be appropriate for the task and hazards present, and they need to fit the wearer properly. Choosing gloves designed to resist exposure to chemical hazards is therefore critical. The following five types of gloves are generally available for workers:
- Leather gloves protect against abrasion.
- Cut-resistant. These gloves can be made from a variety of materials including natural and synthetic fibres and can prevent against cuts, lacerations and in some cases punctures.
- Chemical-resistant.Chemical-resistant glove materials also vary and include things like latex, nitrile and other rubbers and synthetic materials. These gloves can help prevent dermatitis and chemical burns.
- These gloves contain extra layers of protection to keep the hands safe in cold or hot environments (or when touching cold or hot objects).
- Anti-vibration.While vibration is not specifically a skin issue, vibrating tools can cause nerve damage in the hands, and this type of glove can help alleviate that problem.
Two things to assess with chemical hazards
- Think about which chemical you are using and how long a glove will protect you against that chemical.
- Look at how different glove materials will protect against the chemical in question. For example, a neoprene glove might hold up better to a certain chemical than a nitrile glove.
Tip: Consult the chemical’s label and safety data sheet for additional information. Keep in mind that gloves break down over time, so gloves may need to be replaced frequently if chemicals reduce the resistance of a glove.
Sources:
https://www.ccohs.ca/oshanswers/chemicals/how_chem.html
http://health.mo.gov/living/environment/hazsubstancesites/healtheffects.php
https://www.cdc.gov/niosh/topics/skin/default.html
https://www.realsafety.org/2014/08/safety-gloves-and-skin-protection-at-work/
https://www.sunsmart.com.au/downloads/communities/workplaces/protect-your-skin-at-work-brochure.pdf





